Immune Health Profiling in Type 2 Diabetes Mellitus
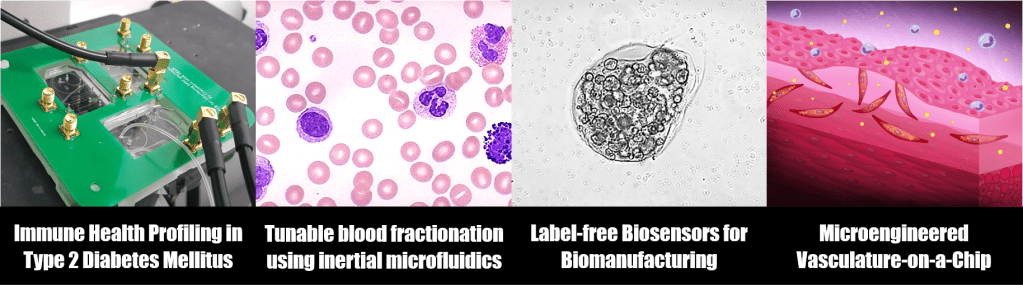
Dysfunction of circulating immune cells (leukocytes) are strongly linked to type 2 diabetes mellitus (T2DM) pathophysiology and atherosclerosis development, but their prognostic potential as inflammatory biomarkers (besides differential cell count) remains largely unexplored due to arduous leukocyte isolation methods. Using a drop of blood, our group has engineered several innovative neutrophil purification microdevices to study neutrophil functions including cell rolling1, chemotaxis2, and NETosis2, and successfully demonstrated their clinical utilities in immune risk stratification of T2DM patients. We have also explored non-traditional single cell biophysical markers including monocyte-platelet aggregates3-4, leukocyte membrane impedance5, as well as developed a “sample in-answer out” integrated microfluidics platform for rapid (~5 min) immune health profiling based on NETosis6.
References
- H.W. Hou, Petchakup, H.M. Tay, Z.Y. Tam, R. Dalan, D.E.K. Chew, K.H.H. Li and B.O. Boehm, Rapid and label-free microfluidic neutrophil purification and phenotyping in diabetes mellitus, Scientific Reports, 2016. 6.
- H.M. Tay, R. Dalan, K.H.H. Li, B.O. Boehm* and H.W. Hou*, A Novel Microdevice for Rapid Neutrophil Purification and Phenotyping in Type 2 Diabetes Mellitus, Small, 2018. 14. 1702832.
- H.M. Tay, W.H. Yeap, R. Dalan, S.C Wong* and H.W. Hou*, Multiplexed Label-Free Fractionation of Peripheral Blood Mononuclear Cells for Identification of Monocyte–Platelet Aggregates, Analytical Chemistry, 2018, 90(24): 14535-14542.
- C. Petchakup, H.M. Tay, W.H. Yeap, R. Dalan, S.C Wong, K.H.H. Li* and H.W. Hou*, Label-free leukocyte sorting and impedance-based profiling for diabetes testing, Biosensors and Bioelectronics, 2018. 118:195-203.
- C. Petchakup, H.M. Tay, K.H.H. Li and H.W. Hou*, Integrated inertial-impedance cytometry for rapid label-free leukocyte isolation and profiling of neutrophil extracellular traps (NETs), Lab on a Chip, 2019. 19, 1736-1746.
Press Articles
- Detecting inflammation in diabetic patients- by News Medical.net, Mediacorp Channel 8
- New NTU kit offers diabetics quick test for inflammation by The Straits Times, Lianhe Zaobao, ScienceDaily
- NTU scientists develop chip that can analyse immune system within minutes by Channel News Asia
- Featured in newspapers Lianhe Zaobao, Lianhe Wanbao
- Scientists build chip to analyze health of white blood cells by Phys Org
- New lab-on-a-chip system quickly identifies health aspects of a person’s immune system by News Medical
Tunable blood fractionation using inertial microfluidics
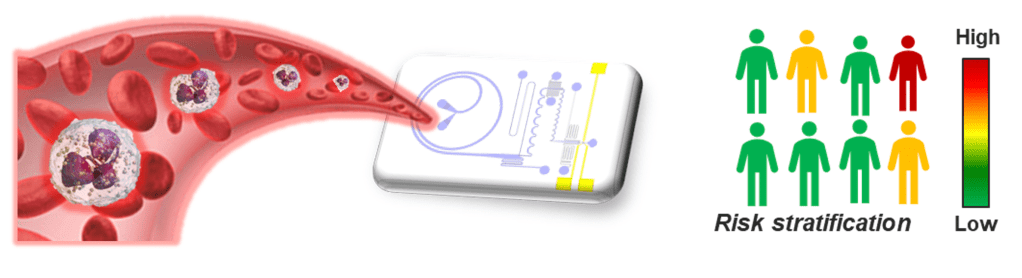
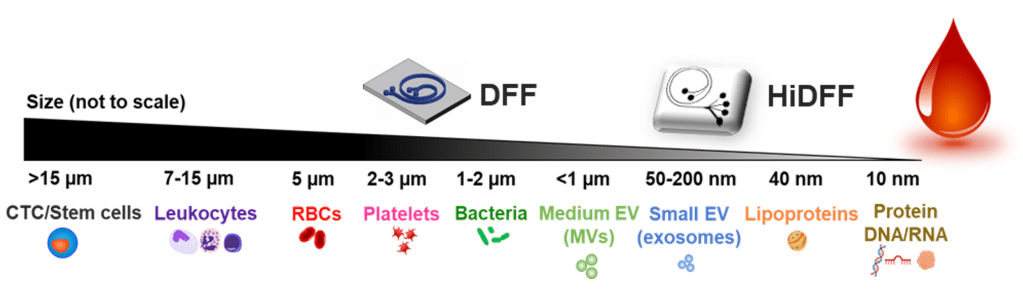
Blood is a highly complex biofluid and most routinely sampled for clinical diagnostics. Ranging from micron-sized cells (WBCs, RBCs) to smaller platelets (cell fragments) and nanoscale cell-derived extracellular vesicles (EV), each component play a critical role in maintaining host defense system, hemostasis, and are implicated in a variety of disease pathophysiology. Hence, there is an unmet critical need to develop novel blood sample preparation tools that can isolate these components based on user need, to advance our understanding in hematology and immunology. Our group has previously pioneered a spiral inertial microfluidics technology, aptly termed as Dean Flow Fractionation (DFF), for separation of micron-sized cells (5 to 30 µm) such as circulating tumor cells1, leukocytes, and stem cells2 from whole blood. Recently, we developed a novel spiral device (High-resolution DFF (HiDFF))3 for sub-micron bacterial sorting and direct isolation of circulating EVs (ExoDFF) from whole blood4. Current research efforts are focused in developing rapid clinical diagnostic solutions based on these technologies.
References
- H.W. Hou, M.E. Warkiani, B.L. Khoo, Z.R. Li, R.A. Soo, D.S.W. Tan, W.T. Lim, J. Han, A.A.S. Bhagat and C.T. Lim, Isolation and retrieval of circulating tumor cells using centrifugal forces. Scientific Reports, 2013. 3.
- D. Yeo, C. Wiraja, Y.Y. Zhou, H.M. Tay, C.J. Xu* and H.W. Hou*, Interference-free micro/nano-particle cell engineering using high-throughput microfluidic separation. ACS Applied Materials & Interfaces, 2015. 7(37): p. 20855-20864.
- H.M. Tay, S. Kharel, R. Dalan, Z.J. Chen, K.K. Tan, B.O. Boehm, S.C.J. Loo and H.W. Hou*, Rapid purification of sub-micrometer particles for enhanced drug release and microvesicles isolation. NPG Asia Materials, 2017. 9: e434.
- H.M. Tay, S.Y. Leong, X. Xu, F. Kong, M. Upadya, R. Dalan, C.Y. Tay, M. Dao, S. Suresh*, H.W Hou*, Direct Isolation of Circulating Extracellular Vesicles from Blood for Vascular Risk Profiling in Type 2 Diabetes Mellitus, Lab on a Chip, In-Press, 2021.
Press Articles
- Spinning Out In Control by Cancerforall
- Capturing Cancer Cells on the Move by TheScientist
- Highly Cited Paper- Top 1% in Clinical Medicine (Web of Science)
Label-free Biosensors for Biomanufacturing
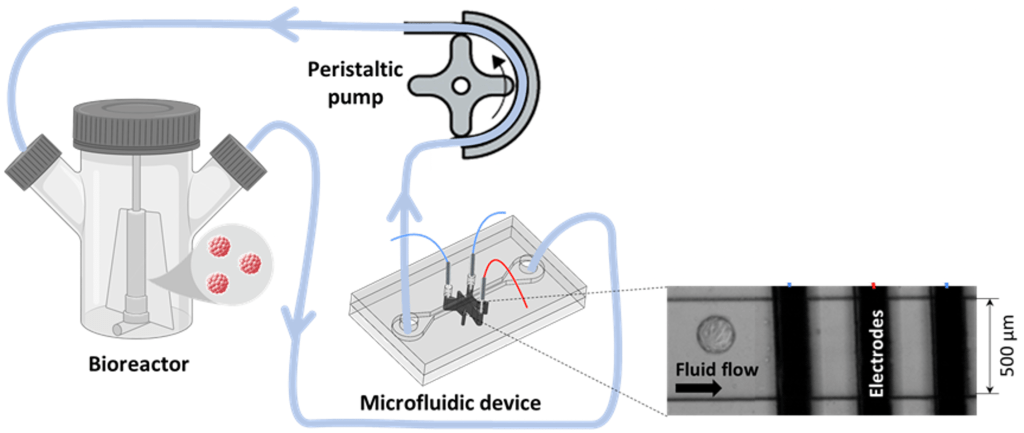
Novel therapeutics using cells (e.g. stem cells and T cells) and extracellular vesicles (EVs) are emerging treatment modalities in many diseases and for personalized medicine. Unlike conventional drugs or biological molecules which have established bioprocessing technologies, cell and EV-based processes require long-term cell culture (~weeks), which advocate a need for non-invasive biosensors to monitor/ control cell quality (growth rate and viability) and EV production. Our group is currently exploring the use of microfluidic-enabled technologies for CART cells profiling1 and biomass monitoring in microcarriers2 with potential applications towards biomanufacturing and tissue engineering for long-term and remote bioprocess monitoring of biomass and cell/EV quality in bioreactors.
References
- C. Petchakup, P.E. Hutchinson, H.M. Tay, S.Y. Leong, K.H.H. Li and H.W. Hou*, Label-free quantitative lymphocyte activation profiling using microfluidic impedance cytometry, Sensors and Actuators B: Chemical, 2021. 339: 129864.
- L. Gong, C. Petchakup, P. Shi, P.L. Tan, L.P. Tan, C.Y. Tay and H.W. Hou*, Direct and label-free cell status monitoring of spheroids and microcarriers using microfluidics impedance cytometry, Small, 2021. 2007500.
Microengineered Vasculature-on-a-Chip
Organ-on-chip platforms are exciting technological tools to study human organ functions and responses at the cellular level. To better understand the pathophysiological impact of immune and vascular dysfunction in metabolic disorders, we are developing biomimetic organ-on-chips to model atherosclerosis, the leading cause of cardiovascular diseases (CVD). As atherosclerosis is a geometrical-focal and multifaceted disease, our research focus on investigating the effects of 1) vascular geometries1, 2) biophysical cues (blood flow)2, 3) platelet and leukocyte-endothelial interactions3, and vascular smooth muscle cells (VSMC) dysfunction4 in atherosclerosis. By serving as surrogate disease models to study host pathophysiological responses in vivo, these biomimetic platforms can significantly advance translational biomedical research in prognostics and diagnostics of CVD, understanding disease etiology and organ physiology, and development of novel therapeutic interventions. The developed organ-on-chip technologies can also be engineered for study of other major disease burden (cancer, infectious diseases) and toxicity testing (pharmacokinetics).

References
- N.V. Menon, H.M. Tay, S.N. Wee, K.H.H. Li and H.W. Hou*, Micro-engineered perfusable 3D vasculature for cardiovascular diseases. Lab on a Chip, 2017. 17(17): p. 2960-2968.
- N.V. Menon, H.M. Tay, K.T. Pang, R. Dalan, S.C. Wong, X.M. Wang, K.H.H. Li and H.W. Hou*, A tunable microfluidic 3D stenosis model to study leukocyte-endothelial interactions in atherosclerosis, APL Bioengineering, 2018. 2(1): 016103.
- N.V. Menon, C.X. Su, K.T. Pang, Z.J. Phua, H.M. Tay, R. Dalan, X.M. Wang, K.H.H. Li and H.W. Hou*, Recapitulating atherogenic flow disturbances and vascular inflammation in a perfusable 3D stenosis model, Biofabrication, 2020. 12(4): 045009.
- C. Su, N.V. Menon, X. Xu, Y.R. Teo, H. Cao, R. Dalan, C.Y. Tay, H.W. Hou*, A novel human arterial wall-on-a-chip to study endothelial inflammation and vascular smooth muscle cell migration in early atherosclerosis, Lab on a Chip, 2021. (in press)
Press Articles
- Featured article in APL Bioengineering (AIP)
- APL Bioengineering Most Downloaded Articles in 2018
- An organ-on-a-chip device that models heart disease by ScienceDaily
- MEMS Chip Aims to Beat Heart Disease by EETimes
- Organ-on-a-chip device successfully models heart disease by Drug target review